The necessary conduct: Exploratory multiregional clinical trials in East Asia - Jeon - 2021 - Clinical and Translational Science - Wiley Online Library

Amazon.co.jp: Multiregional Clinical Trials for Simultaneous Global New Drug Development (Chapman & Hall/CRC Biostatistics Series) : Chen, Joshua, Quan, Hui: Foreign Language Books

Advancing International Pediatric Clinical Research Webinar Series - The Multi-Regional Clinical Trials Center of Brigham and Women's Hospital and Harvard

Clinical Data Interchange Standards Consortium (CDISC) and the Multi-Regional Clinical Trials Center of Brigham and Women's Hospital (MRCT Center) Collaborate to Offer Plain Language Clinical Research Definitions as a New Standard

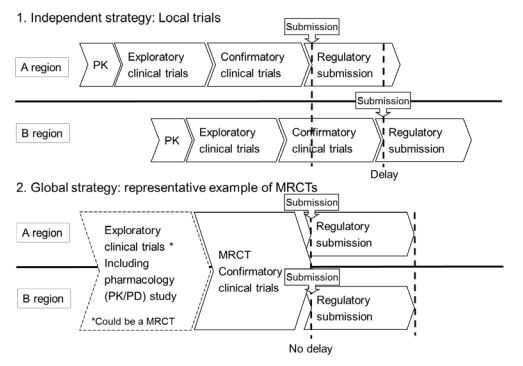
The Current Status of “Multi-Regional Clinical Trials (MRCT)” ~ <br><span> </span> What we should do now?|Global Clinical Research Support Group

Strategic inclusion of regions in multiregional clinical trials - Seung Yeon Song, Deborah Chee, EunYoung Kim, 2019

Clinical Data Interchange Standards Consortium (CDISC) and the Multi-Regional Clinical Trials Center of Brigham and Women's Hospital (MRCT Center) Collaborate to Offer Plain Language Clinical Research Definitions as a New Standard