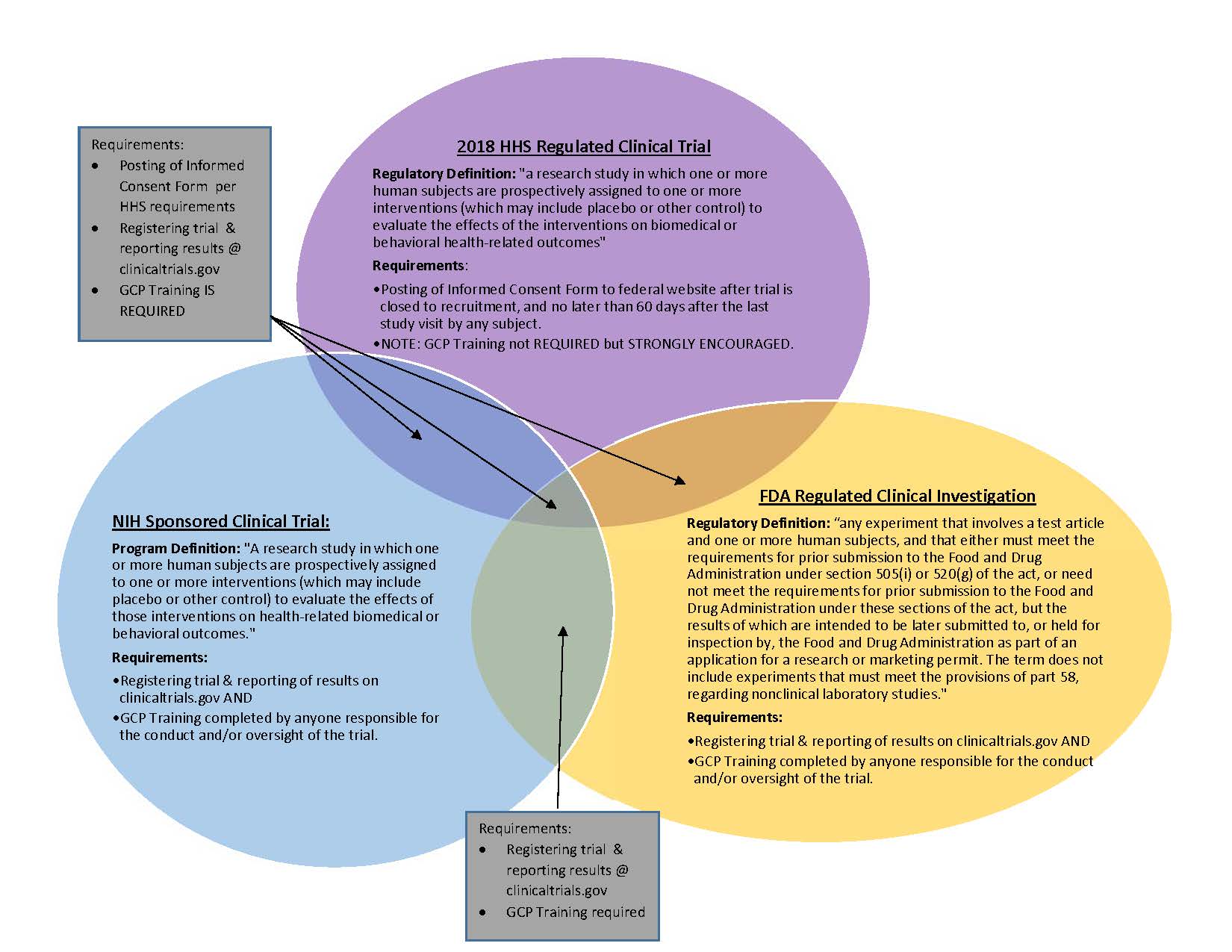
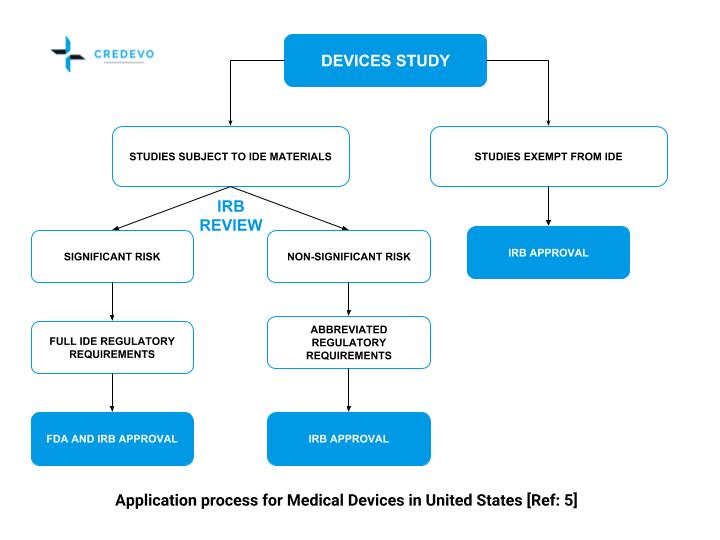
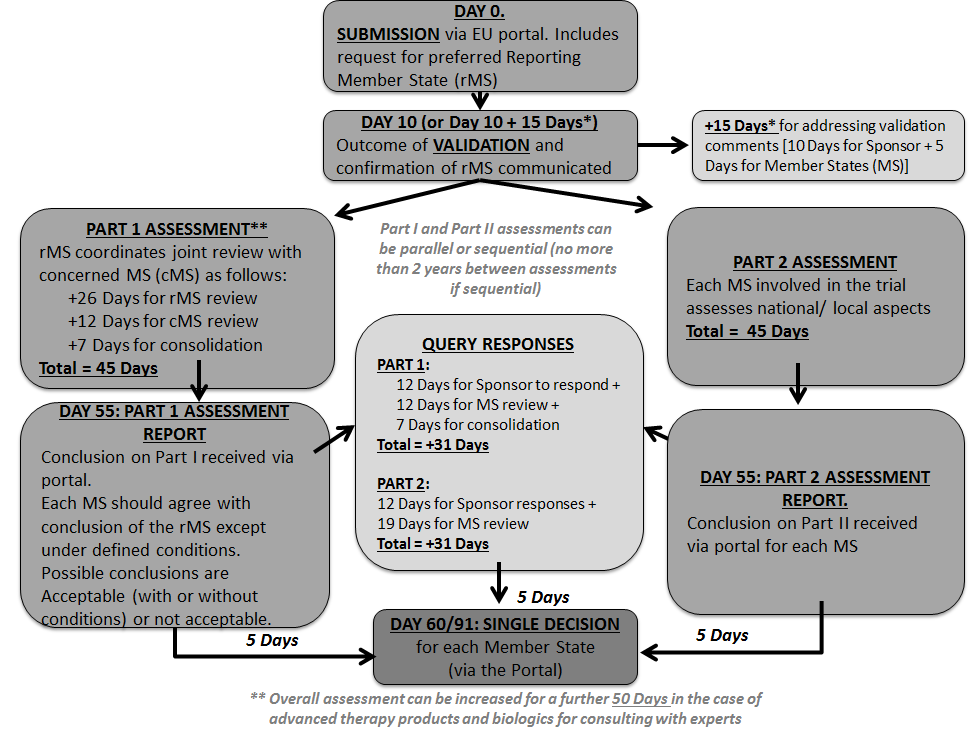
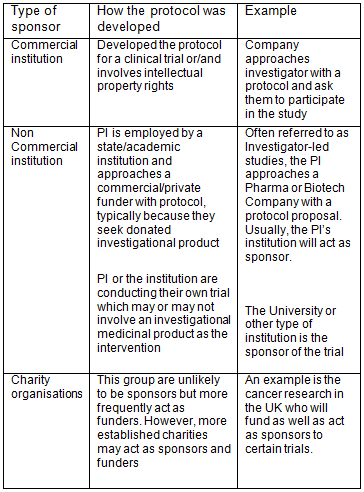
Guidance Document: Part C, Division 5 of the Food and Drug Regulations “Drugs for Clinical Trials Involving Human Subjects” (GUI-0100) - Canada.ca
Issues, challenges, and the way forward in conducting clinical trials among neonates: investigators' perspective | Journal of Perinatology
Issues, challenges, and the way forward in conducting clinical trials among neonates: investigators' perspective | Journal of Perinatology

More than a box to check: Research sponsor and clinical investigator perspectives on making GCP training relevant - ScienceDirect

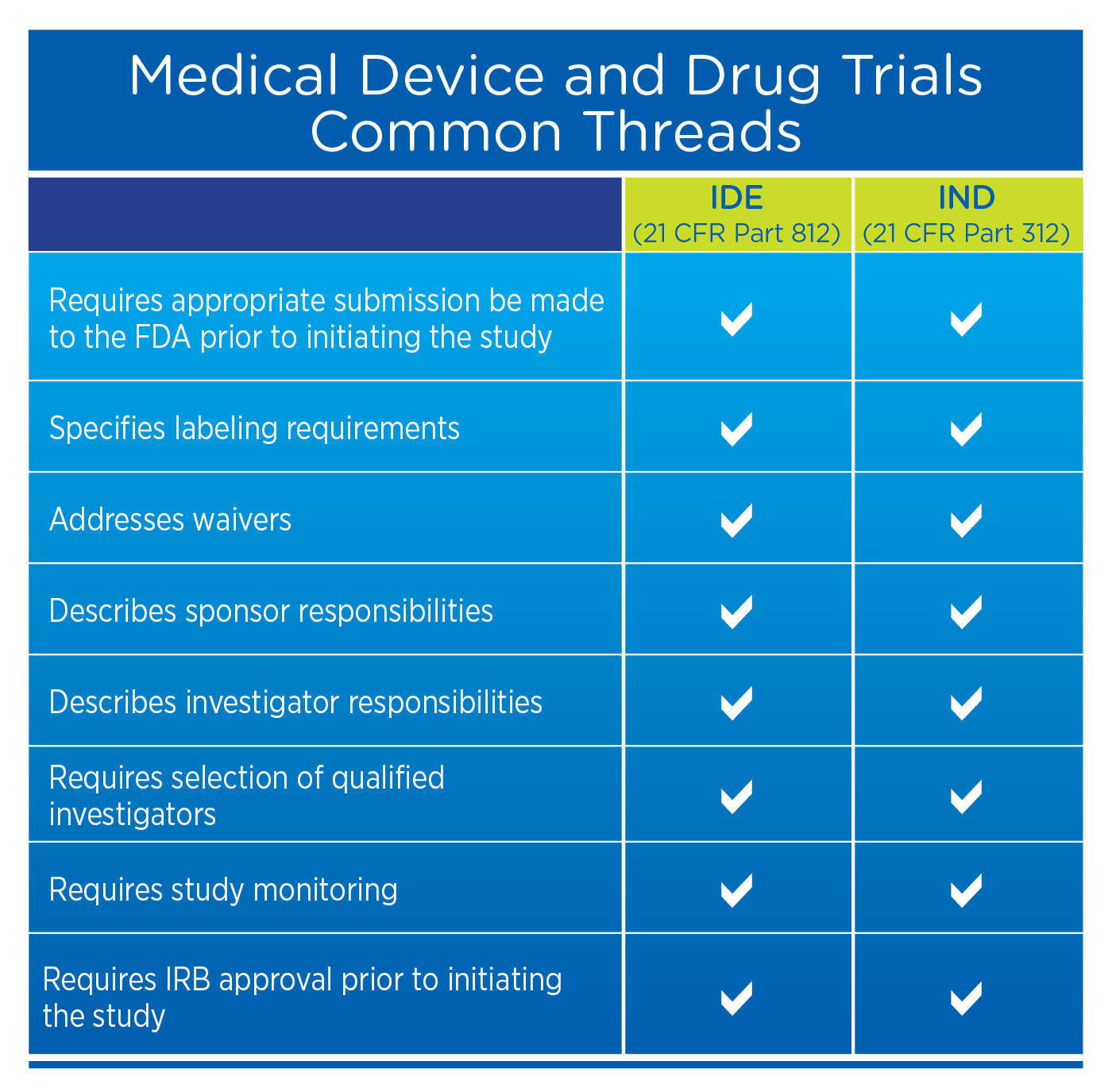
Regulatory Approval – Clinical Trial Medical Monitoring Plan | Online Clinical Research Courses In India
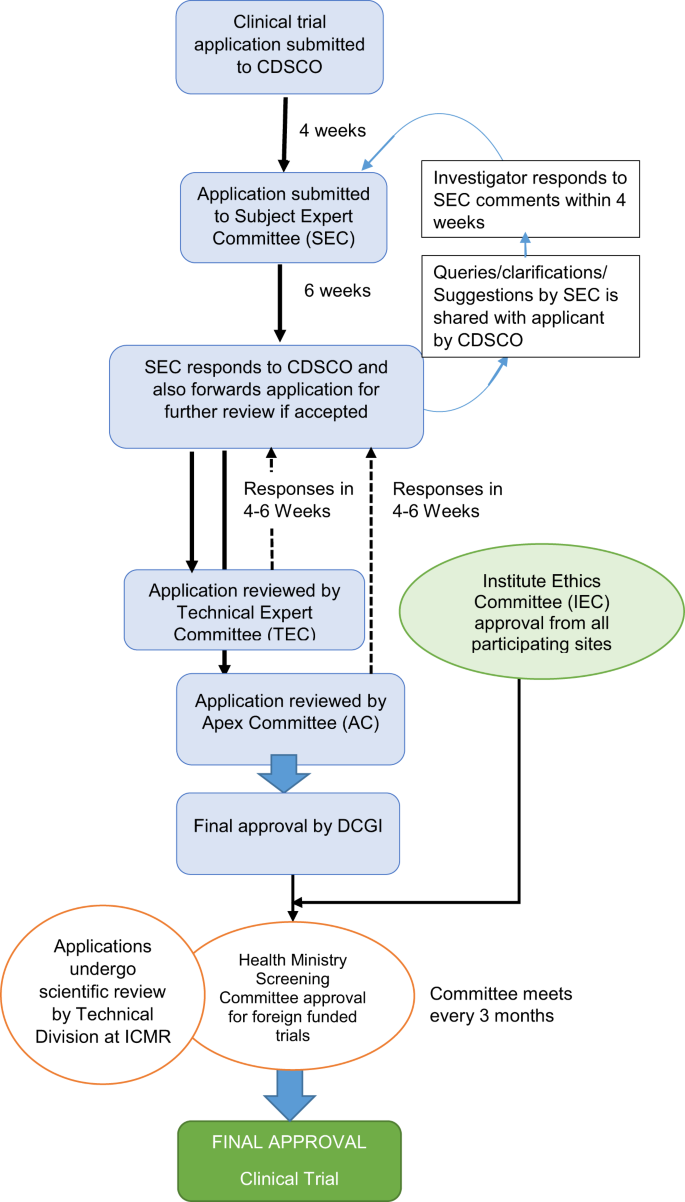
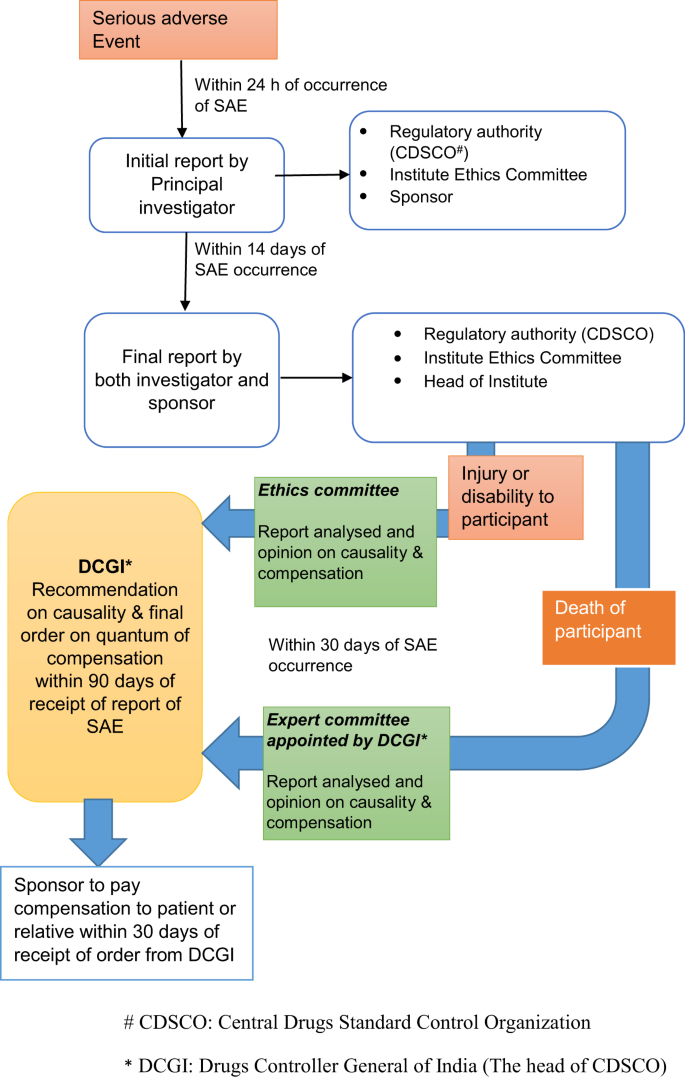
![Guidance for Clinical Trial Sponsors Clinical Trial Applications · PDF file 2018-09-16 · GUIDANCE FOR CLINICAL TRIAL SPONSORS Clinical Trial Applications Published by authority - [PDF Document] Guidance for Clinical Trial Sponsors Clinical Trial Applications · PDF file 2018-09-16 · GUIDANCE FOR CLINICAL TRIAL SPONSORS Clinical Trial Applications Published by authority - [PDF Document]](https://cdn.vdocument.in/doc/1200x630/5e9e775a7de2094ce32c3542/guidance-for-clinical-trial-sponsors-clinical-trial-applications-2018-09-16-guidance.jpg?t=1651530030)
Guidance for Clinical Trial Sponsors Clinical Trial Applications · PDF file 2018-09-16 · GUIDANCE FOR CLINICAL TRIAL SPONSORS Clinical Trial Applications Published by authority - [PDF Document]

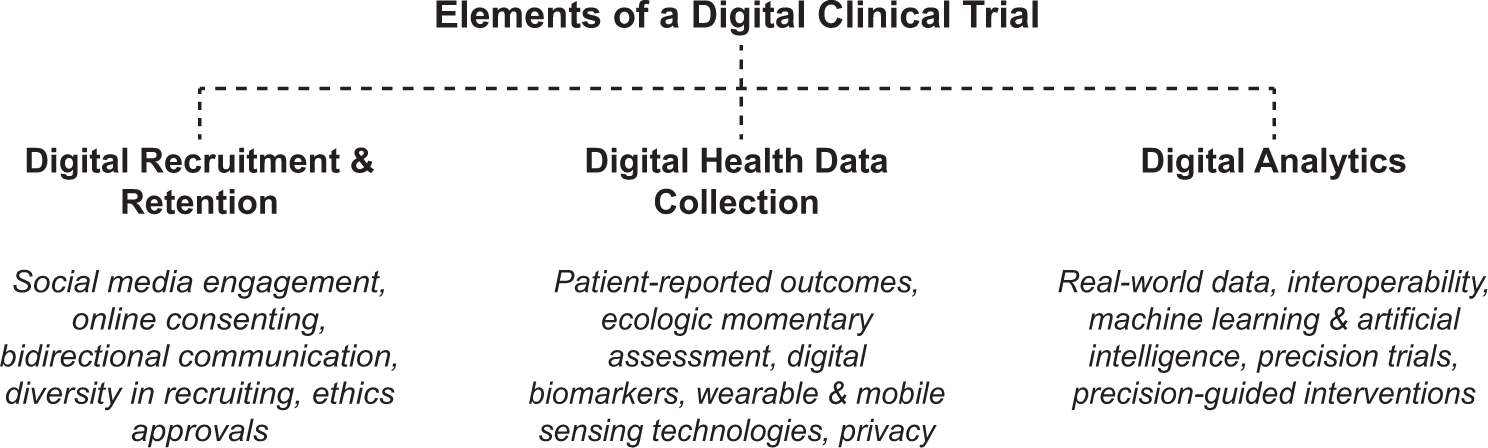
Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension - The Lancet Digital Health
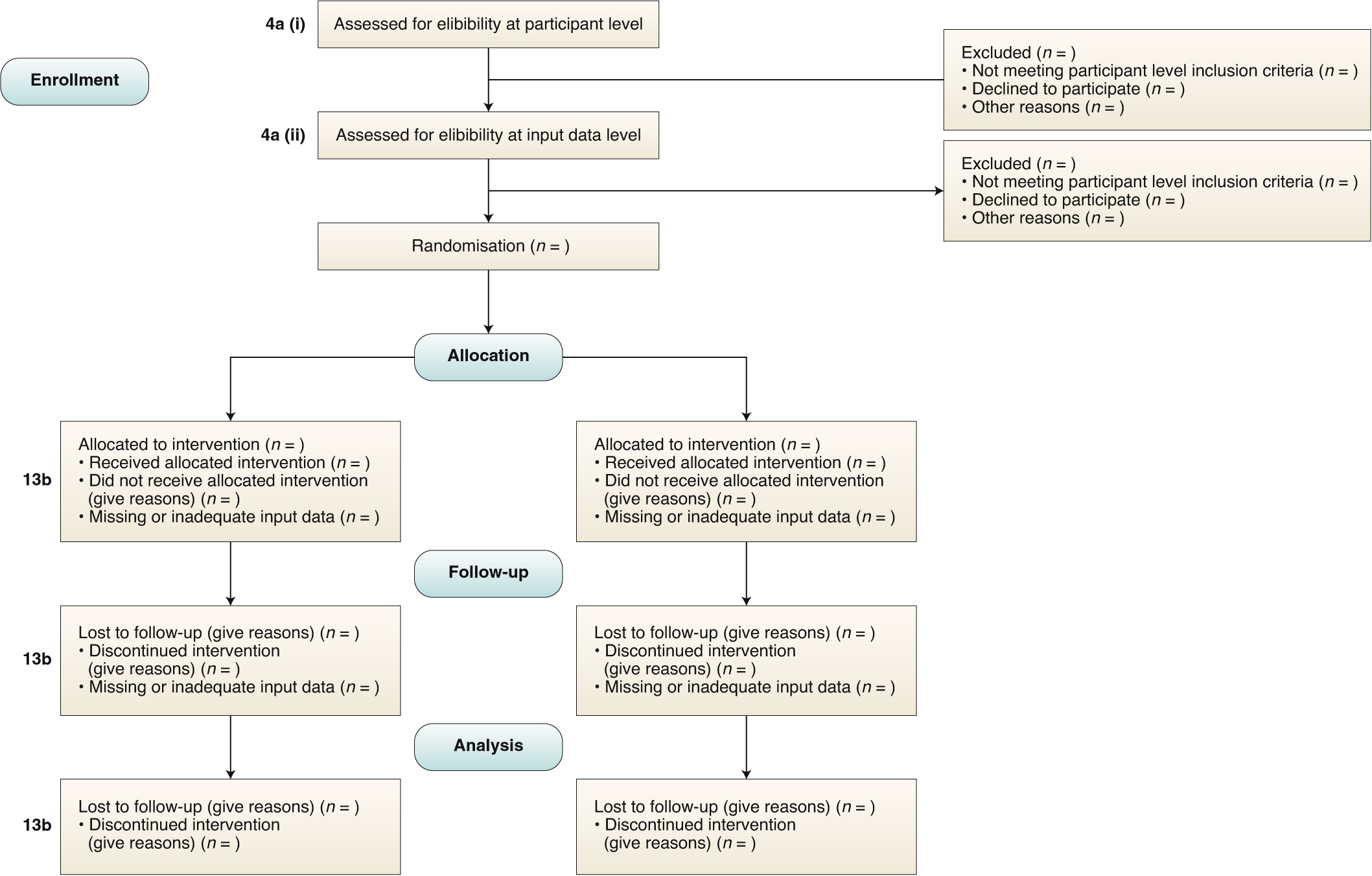
Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension | Nature Medicine